


Next: 4. Basic GC Theory
Up: 3.3 Liquid-Liquid Extraction
Previous: 3.3.3 Applying acid-base extraction
Contents
- The pH of the aqueous phase should not be made too low or too high.
Extreme pH's may cause undesired chemical reactions. Only a change
to or from a salt form is required. In particular, the pK
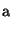 or pK
or pK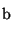 of the substance being converted determines
the target pH.
of the substance being converted determines
the target pH.
- More complex mixtures than the one in Section 3.3.3
can be separated by subsequent pH manipulations.
- Some organic bases (phenethylamines, for example) do not crystallize
well (or at all) which may require conversion to the salt form after
extraction. The procaine in Section 3.3.3
is also an example.
- Some materials have multiple crystal forms (polymorphism). Each crystal
form will have a different IR spectrum; therefore, in these cases,
it is important to crystallize from the same solvent used to obtain
standard or library spectra.
John S. Riley, DSB Scientific Consulting